Removing H2S from Gas Streams
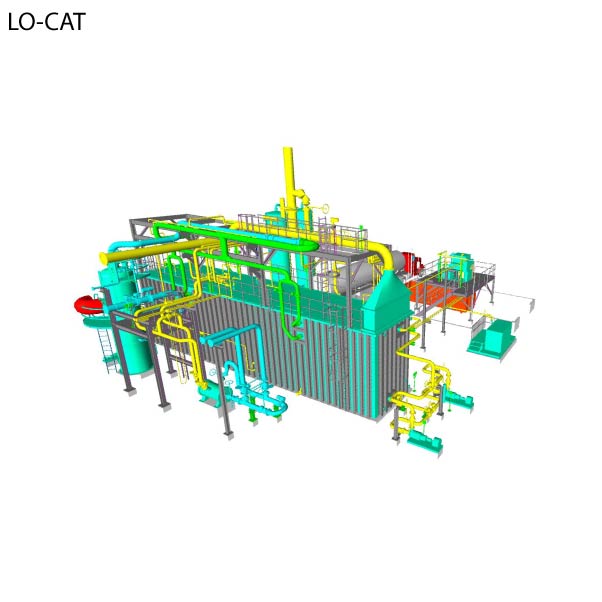
LO-CAT® | Sulfur Recovery Technology
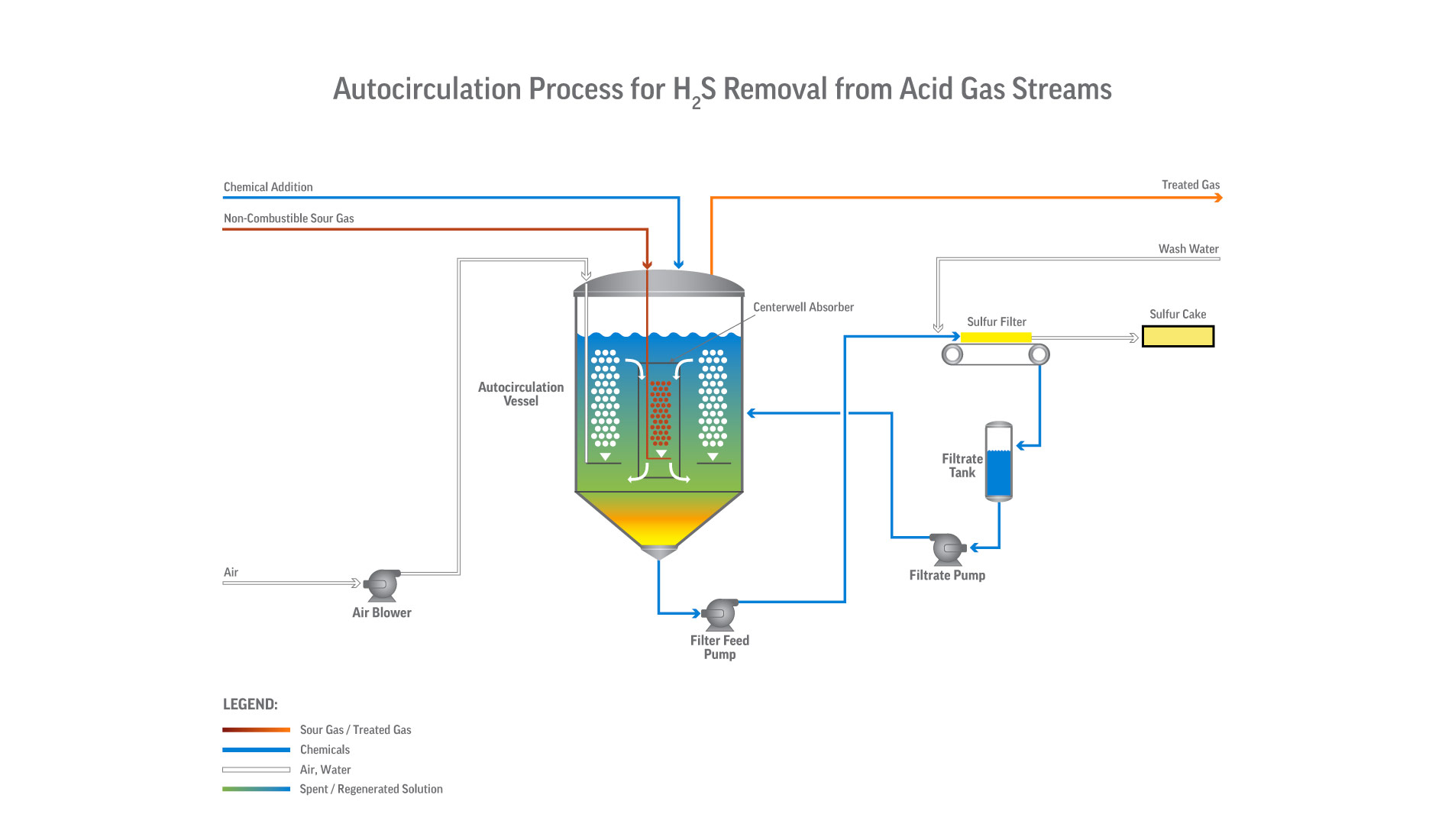
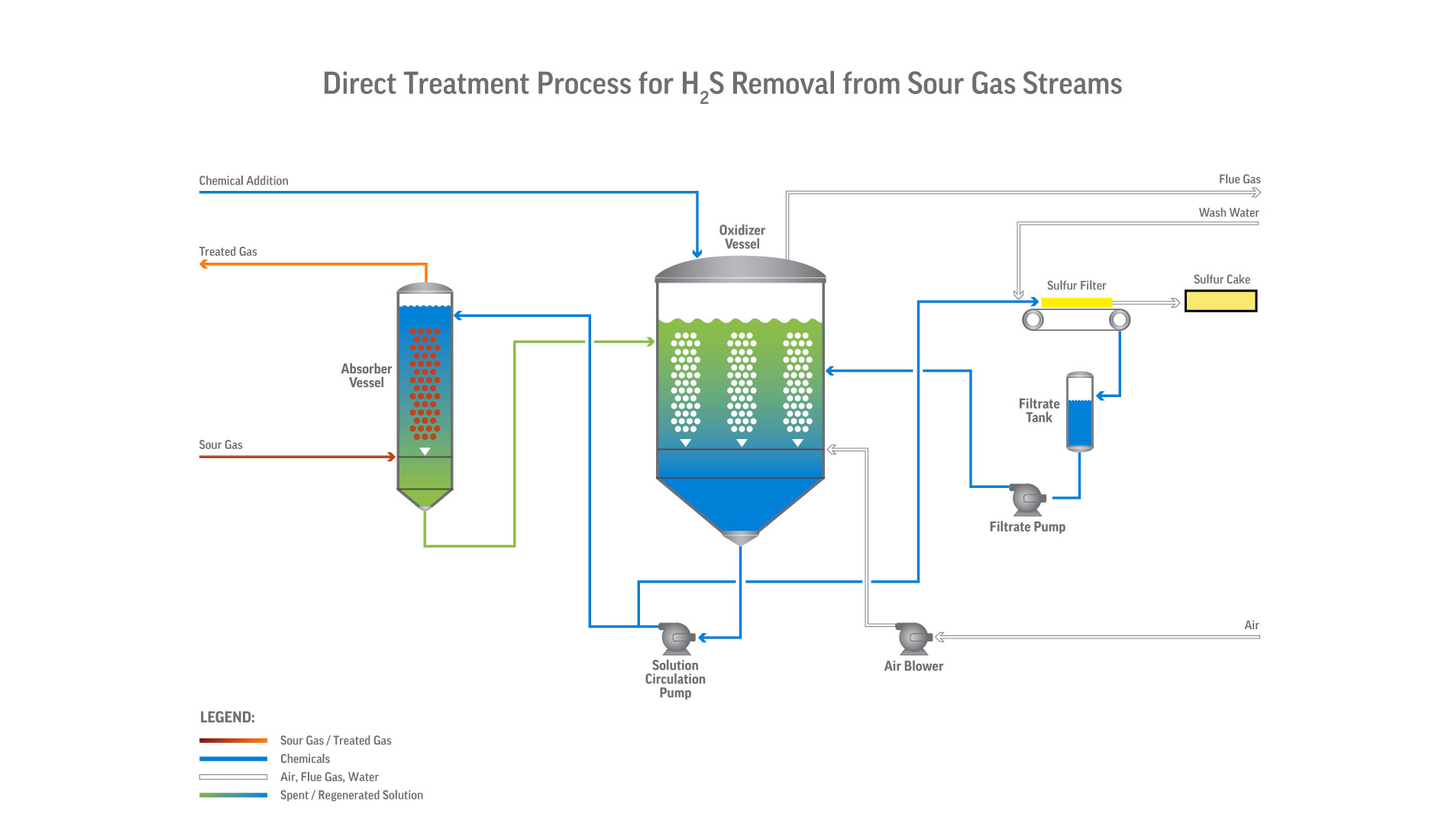
With Merichem’s exclusive sulfur recovery technology, this patented liquid redox system uses a chelated iron solution to convert H2S into innocuous, elemental sulfur.
LO-CAT® Advantages
- Can remove H2S from virtually any gas stream
- Available process guarantees less than 1 ppm H2S
- Chemical-based sulfur removal — more reliable and flexible than biological treatment
- No liquid waste streams
- No hazardous waste products produced
- High turndown (up to 100%)
- Highly customizable to specific treatment needs
- Aqueous-based process at low temperatures
- Uses proprietary catalyst solution and chemicals
- Low operating cost
- 99.9 % or more reduction of H2S
- Continuously regenerated catalyst for low operating costs
- Lower OPEX vs. scavengers; lower CAPEX vs. Claus plants
- Reliable and cost-efficient removal of H2S; easily accommodates sulfur loads up to 20 tons/day
- Process can directly treat gas streams or serve as the sulfur recovery unit (SRU) downstream of an acid-gas removal (amine) unit
Innovations for Performance
Merichem® has provided innovative technology and equipment to the oil and gas industry for over 40 years. Our proven solutions have continuously helped our customers achieve greater performance and profitability.
Comprehensive and Committed
Complementing our depth of expertise in gas processing is a breadth of capability that includes engineering, design, fabrication, analytics, testing and service support. Merichem® is here to help our customers meet their specifications, performance and warranty requirements.